MOHAP Product Classification & Registration in UAE
If you operate in the healthcare, pharmaceutical, or medical device industry, obtaining approval from the UAE Ministry of Health and Prevention (MOHAP) is essential before any product can enter the market. From medical device classification to full MOHAP registration in the UAE, every healthcare product must meet strict regulatory requirements.
Nextmove provides expert support for MOHAP product registration, medical device classification, and regulatory licensing, ensuring your healthcare products comply with UAE healthcare regulations. Our team guides manufacturers, importers, and distributors through the entire approval process — helping you secure MOHAP regulatory approval efficiently and without unnecessary delays.
With our dedicated MOHAP registration services in the UAE, businesses can avoid costly mistakes, documentation errors, and long approval times. We manage the full process from product evaluation and medical device classification to final submission through the official MOHAP regulatory system, helping your products enter the UAE healthcare market faster.
MOHAP Compliance
We ensure your medical device registration in the UAE meets all regulatory requirements set by the UAE Ministry of Health and Prevention (MOHAP). Our specialists handle compliance checks, regulatory documentation, and submission processes to secure official approval.
Full-Service Support
Our team provides end-to-end assistance for MOHAP product registration, supporting manufacturers, importers, and distributors at every stage — from product classification and documentation preparation to regulatory submission and approval.
Wide Product Range
Our regulatory expertise covers a wide range of healthcare products, including medical devices, diagnostic equipment, pharmaceuticals, and healthcare technologies, ensuring smooth MOHAP regulatory approval in the UAE healthcare market.
Achieving Your Vision
Planning for retirement is essential to your long-term financial well-being. At Finovate, our experienced team collaborates with you to identify your retirement goals and crafts a tailored, comprehensive strategy to help you achieve them with confidence.
What we do

Import Clearance Support
Our MOHAP import clearance service helps healthcare companies obtain the required permits for importing medical devices and pharmaceutical products into the UAE. We assist with regulatory documentation, customs approvals, and compliance with Ministry of Health and Prevention requirements.
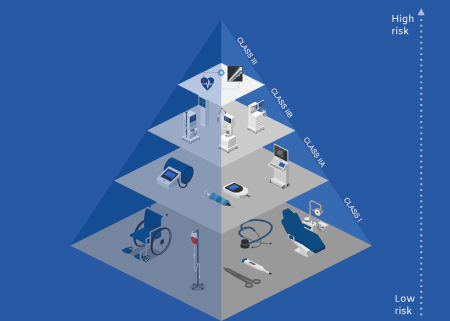
Medical
Device
Classification
Medical device classification is a critical step in the MOHAP registration process. Devices are categorized based on risk levels to determine the regulatory pathway required for approval. Our experts help companies identify the correct classification to ensure smooth product registration.
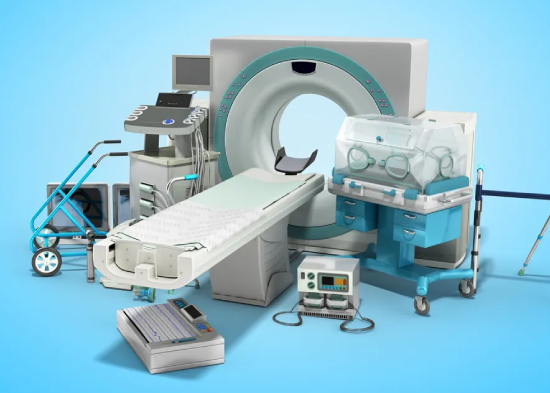
Medical
Device
Registration
Medical device registration with MOHAP ensures healthcare products meet UAE safety and regulatory standards. We assist manufacturers and distributors with documentation preparation, regulatory submissions, and compliance to obtain market approval.

Authorized Representative Services
Foreign manufacturers must appoint an authorized representative in the UAE for MOHAP medical device registration. Our service ensures regulatory communication, documentation management, and compliance with UAE healthcare authority requirements.
See What Our Clients Are Saying
“I hired Finovate for a small project & was very happy. He not only answered all my questions, but he didn’t treat me like a “small project”.
I was very satisfied & would recommend.”
“Finovate has been instrumental in our growth. Their team took the time to truly understand our needs and helped us eliminate inefficiencies.”
“Partnering with Finovate was a game-changer for us. They took the time to understand our challenges and helped us streamline our operations for success.”




